Abstract
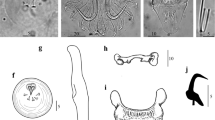
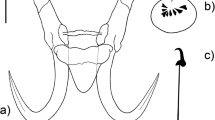
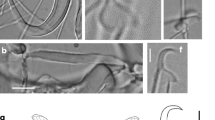
Eight new species of Gyrodactylus are described from Poecilia mexicana, Poeciliopsis gracilis, Pseudoxiphophorus bimaculatus [syn. = Heterandria bimaculata], and Xiphophorus hellerii collected in the Nautla and La Antigua River Basins in Veracruz, and in the Tecolutla River Basin in Puebla, Mexico. Analyzing the morphology of the marginal hooks, Gyrodactylus pseudobullatarudis n. sp. and Gyrodactylus xtachuna n. sp. are both very similar to Gyrodactylus bullatarudis; Gyrodactylus takoke n. sp. resembles Gyrodactylus xalapensis; Gyrodactylus lhkahuili n. sp. is similar to Gyrodactylus jarocho; and both Gyrodactylus microdactylus n. sp. and Gyrodactylus actzu n. sp. are similar to Gyrodactylus poeciliae in that all three species possess extremely short shaft points. A hypothesis of the systematic relationships of the eight new Gyrodactylus species and some of the known gyrodactylids infecting poeciliids was constructed with sequences of the Internal Transcribed Spacers (ITS1 and ITS2) and the 5.8S ribosomal gene of the rRNA. Phylogenetic trees showed that the new and previously described species of Gyrodactylus infecting poeciliid fishes do not form a monophyletic assemblage. Trees also showed that the eight new species described morphologically correspond to well-supported monophyletic groups; and that morphologically similar species are also phylogenetically close. Additionally, we correct previous erroneous records of the presence of Gyrodactylus bullatarudis on wild Poecilia mexicana and Xiphophorus hellerii collected in Mexico, as re-examination of the original specimens indicated that these corresponded to Gyrodactylus pseudobullatarudis n. sp. (infecting Poecilia mexicana and Xiphophorus hellerii) and to Gyrodactylus xtachuna n. sp. (on Xiphophorus hellerii). Finally, given the widespread anthropogenic translocation of poeciliid fishes for the aquarium trade and mosquito control programs, as well as the existence of invasive, feral poeciliid populations worldwide, we discuss the possibility that gyrodactylid parasites could be introduced along with the fish hosts—this work provides taxonomic information to assess that possibility, as it describes parasites collected from poeciliid fishes within their native distribution range.









Similar content being viewed by others
References
Agorreta A, Domínguez-Domínguez O, Reina RG, Miranda R, Bermingham E, Doadrio I (2013) Phylogenetic relationships and biogeography of Pseudoxiphophorus (Teleostei: Poecilidae) based on mitochondrial and nuclear genes. Mol Phylogenet Evol 66:80–90
An L, Jara CA, Cone DK (1991) Five species of Gyrodactylus Nordmann, 1832 (Monogenea) from fresh-water fishes of Peru. Can J Zool 69:1199–1202
Cable J, van Oosterhout C, Barson N, Harris PD (2005) Gyrodactylus pictae n. sp. (Monogenea: Gyrodactylidae) from the Trinidadian swamp guppy Poecilia picta Regan, with a discussion on species of Gyrodactylus von Nordmann, 1832 and their poeciliid hosts. Syst Parasitol 60:159–164
Cable J, Archard GA, Mohammed RS, McMullan M, Stephenson JF, Hansen H, van Oosterhout C (2013) Can parasites use predators to spread between primary hosts? Parasitology 140:1–6
Cribb TH, Chisholm LA, Bray RA (2002) Diversity in the Monogenea and Digenea: does lifestyle matter? Int J Parasitol 32:321–328
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772
Dove ADM (2000) Richness patterns in the parasite communities of exotic poeciliid fishes. Parasitology 120:609–623
Dove ADM, Ernst I (1998) Concurrent invaders—four exotic species of Monogenea now established on exotic freshwater fishes in Australia. Int J Parasitol 28:1755–1764
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Galtier N, Gouy M, Gautier C (1996) SEAVIEW and PHYLO_WIN: two graphic tools for sequence alignment and molecular phylogeny. Comput Appl Biosci 12:543–548
García-Vásquez A, Hansen H, Christison KW, Bron JE, Shinn AP (2011) Description of three new species of Gyrodactylus Nordmann 1832 (Monogenea) from oreochromids (Oreochromis, Cichlidae). Acta Parasitol 56:20–33
Gilmore SR, Cone DK, Lowe G, King SK, Jones SRM, Abbott CL (2012) Molecular phylogeny of Gyrodactylus (Monogenea) parasitizing fishes in fresh water, estuarine and marine habitats in Canada. Can J Zool 90:776–786
Guindon S, Gascuel O (2003) A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Syst Biol 52:696–704
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Harris PD (1986) Species of Gyrodactylus von Nordmann, 1832 (Monogenea: Gyrodactylidae) from poeciliid fishes, with a description of G. turnbulli sp. nov. from the guppy, Poecilia reticulata Peters. J Nat Hist 20:183–191
Harris PD, Cable J (2000) Gyrodactylus poeciliae n. sp. and G. milleri n. sp. (Monogenea: Gyrodactylidae) from Poecilia caucana (Steindachner) in Venezuela. Syst Parasitol 47:79–85
Harris PD, Lyles AM (1992) Infections of Gyrodactylus bullatarudis and Gyrodactylus turnbulli on Guppies (Poecilia reticulata) in Trinidad. J Parasitol 78:912–914
Harris PD, Shinn AP, Cable J, Bakke TA (2004) Nominal species of the genus Gyrodactylus von Nordmann 1832 (Monogenea: Gyrodactylidae), with a list of principal host species. Syst Parasitol 59:1–27
Harris PD, Shinn AP, Cable J, Bakke TA, Bron J (2008) GyroDb: gyrodactylid monogeneans on the web. Trends Parasitol 24:109–111
Hoberg EP, Agosta SJ, Boeger WA, Brooks DR (2015) An integrated parasitology: revealing the elephant through tradition and invention. Trends Parasitol 31:128–133
Holitzki TM, Mackenzie RA, Wiegner TN, McDermid KJ (2013) Differences in ecological structure, function, and native species abundance between native and invaded Hawaiian streams. Ecol Appl 23:1367–1383
Huyse T, Volckaert FAM (2002) Identification of a host-associated species complex using molecular and morphometric analyses, with the description of Gyrodactylus rugiensoides n. sp. (Gyrodactylidae, Monogenea). Int J Parasitol 32:907–919
Huyse T, Volckaert FAM (2005) Comparing host and parasite phylogenies: Gyrodactylus flatworms jumping from goby to goby. Syst Biol 54:710–718
Kritsky DC, Fritts TH (1970) Monogenetic trematodes from Costa Rica with the proposal of Anacanthocotyle gen. n. (Gyrodactylidae: Isancistrinae). Proc Helmithol Soc Wash 37:63–68
Kvach Y, Kornyychuk Y, Mierzejewska K, Rubtsova N, Yurakhno V, Grabowska J, Ovcharenko M (2014) Parasitization of invasive gobiids in the eastern part of the Central trans-European corridor of invasion of Ponto-Caspian hydrobionts. Parasitol Res 113:1605–1624
Lucký Z (1973) Gyrodactylus rasini n. sp. (Monogenoidea: Gyrodactylidae) a parasite on the gills of Xiphophorus hellerii bred as an aquarium fish in Czechoslovakia. Vet Med 18:647–652
Matějusova I, Gelnar M, McBeath AJA, Collins CM, Cunningham CO (2001) Molecular markers for gyrodactylids (Gyrodactylidae: Monogenea) from five fish families (Teleostei). Int J Parasitol 31:738–745
Miller RR (2005) Freshwater fishes of Mexico. The University of Chicago Press, USA
Morales-Cazan A, Albert JS (2012) Monophyly of Heterandriini (Teleostei: Poeciliidae) revisited: a critical review of the data. Neotrop Ichthyol 10:19–44
Ondračková M, Dávidová M, Pečinková M, Blažek R, Gelnar M, Valová Z, Černý J, Jurajda P (2005) Metazoan parasites of Neogobius fishes in the Slovak section of the River Danube. J Appl Ichthyol 21:345–349
Paperna I (1968) Monogenetic trematodes collected from freshwater fish in Ghana. Second report. Bamidgeh 20:88–90
Pérez-Ponce de León G, Nadler SA (2010) What we don’t recognize can hurt us: a plea for awareness about cryptic species. J Parasitol 96:453–464
Pyke GH (2008) Plague minnow or mosquito fish? A review of the biology and impacts of introduced Gambusia species. Annu Rev Ecol Evol Syst 39:171–191
Razo-Mendivil U, Vázquez-Domínguez E, Rosas-Valdez R, Pérez-Ponce de León G, Nadler SA (2010) Phylogenetic analysis of nuclear and mitochondrial DNA reveals a complex of cryptic species in Crassicutis cichlasomae (Digenea: Apocreadiidae), a parasite of Middle-American cichlids. Int J Parasitol 40:471–486
Razo-Mendivil U, Rosas Valdez R, Rubio-Godoy M, Pérez-Ponce de León G (2015) The use of mitochondrial and nuclear sequences in prospecting for cryptic species in Tabascotrema verai (Digenea: Cryptogonomidae), a parasite of Petenia splendida (Cichlidae) in Middle America. Parasitol Int 64:173–181
Rogers WA, Wellborn TL Jr (1965) Studies on Gyrodactylus (Trematoda: Monogenea) with descriptions of five new species from the southeastern U. S. J Parasitol 51:977–982
Ronquist F, Teslenko M, van der Mark P, Ayres D, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Rubio-Godoy M, Paladini G, García-Vásquez A, Shinn AP (2010) Gyrodactylus jarocho n. sp. and Gyrodactylus xalapensis n. sp. (Platyhelminthes: Monogenea) from Mexican poeciliids (Teleostei: Cyprinodontiformes), with comments on the known gyrodactylid fauna infecting poeciliid fish. Zootaxa 2509:1–29
Rubio-Godoy M, Paladini G, Freeman M, García-Vásquez A, Shinn AP (2012) Morphological and molecular characterisation of Gyrodactylus salmonis (Platyhelminthes, Monogenea) isolates collected in Mexico from rainbow trout (Oncorhynchus mykiss Walbaum). Vet Parasitol 186:289–300
Salgado-Maldonado G, Rubio-Godoy M (2014) Helmintos parásitos de peces de agua dulce introducidos. In: Koleff P, Mendoza R (eds) Especies acuáticas invasoras en México. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, Mexico City, pp 269–285
Salgado-Maldonado G, Cabañas-Carranza G, Caspeta-Mandujano JM, Soto-Galera E, Mayén-Peña E, Brailovsky D, Báez-Valé R (2001) Helminth parasites of freshwater fishes of the Balsas River drainage basin southwestern Mexico. Comp Parasitol 68:196–203
Salgado-Maldonado G, Aguilar-Aguilar R, Cabañas-Carranza G, Soto-Galera E, Mendoza-Palmero C (2005) Helminth parasites in freshwater fish from the Papaloapan river basin, Mexico. Parasitol Res 96:69–89
Salgado-Maldonado G, Novelo-Turcotte MT, Vázquez G, Caspeta-Mandujano JM, Quiroz-Martínez B, Favila M (2014) The communities of helminth parasites of Heterandria bimaculata (Teleostei: Poeciliidae) from the upper Río La Antigua basin, east-central Mexico show a predictable structure. Parasitology 141:970–980
Schwarz G (1978) Estimating the dimension of a model. Ann Stat 6:461–464
Shinn AP, Harris PD, Cable J, Bakke TA, Paladini G, Bron JE (2011) GyroDb. World Wide Web electronic publication. (http://www.gyrodb.net)
Swofford DL (2003) PAUP*. Phylogenetic Analysis using Parsimony (*and other Methods), version 4.0b10. Sinauer Associates, Sunderland
Turnbull ER (1956) Gyrodactylus bullatarudis n. sp. from Lebistes reticulatus Peters with a study of its life-cycle. Can J Zool 34:583–594
Valero A, Macías-García C, Magurran AE (2008) Heterospecific harassment of native endangered fishes by invasive guppies in Mexico. Biol Lett 4:149–152
van Oosterhout C, Mohammed RS, Hansen H, Archard GA, McMullan M, Weese DJ, Cable J (2007) Selection by parasites in spate conditions in wild Trinidadian guppies (Poecilia reticulata). Int J Parasitol 37:805–812
Vanhove MPM, Economou AN, Zogaris S, Giakoumi S, Zanella D, Volckaert FAM, Huyse T (2014) The Gyrodactylus (Monogenea, Gyrodactylidae) parasite fauna of freshwater sand gobies (Teleostei, Gobioidei) in their centre of endemism, with description of seven new species. Parasitol Res 113:653–668
Xavier R, Faria PJ, Paladini G, van Oosterhout C, Johnson M, Cable J (2015) Evidence for cryptic speciation in directly transmitted gyrodactylid parasites of Trinidadian guppies. PLoS ONE 10:e0117096
Ziętara MS, Lumme J (2002) Speciation by host switch and adaptive radiation in a fish parasite genus Gyrodactylus (Monogenea, Gyrodactylidae). Evolution 56:2445–2458
Zwickl DJ (2006) Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maximum likelihood criterion. Ph.D. thesis: The University of Texas at Austin, USA
Acknowledgments
We are grateful to Ismael Guzmán (INECOL) for help collecting hosts and worms; Martha Salazar and Cutperto Mora for the collection of fish specimens in Puebla; Diego Santiago Alarcón (INECOL) for the loan of the microscope to take microphotographs; and Luis García Prieto from the Colección Nacional de Helmintos (CNHE), Universidad Nacional Autónoma de México (UNAM) for the loan of Gyrodactylus type material. We thank Mark Freeman (University of Malaya, Malaysia) for designing the primers ITS1-fm and ITS2-rm, and for allowing us to publish the ITS sequences of Gyrodactylus jarocho and Gyrodactylus xalapensis, which he obtained. We thank Germán Muñoz, Mario Garduño and Héctor Basurto (all UNAM) for allowing us to work at Rancho El Clarín, Tlapacoyan. Field sampling was performed according to Mexican codes of practice and received approval of the Mexican Ministry of the Environment and Natural Resources (SEMARNAT) under scientific collection permit SGPA/DGVS/02967/14. This research was supported by grants to MRG from Consejo Nacional de Ciencia y Tecnología (CONACyT no. CB 168306) and Fundación Produce Veracruz/Red Nacional de Información e Investigación en Pesca y Acuacultura (FUNPROVER/RNIIPA). AGV thanks CONACyT for her postdoctoral fellowship (Becario 20340). This study was presented at the XIII ICOPA 2014 conference.
Author information
Authors and Affiliations
Corresponding author
Additional information
Adriana García-Vásquez and Ulises Razo-Mendivil are the principal authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
(DOCX 74 kb)
Rights and permissions
About this article
Cite this article
García-Vásquez, A., Razo-Mendivil, U. & Rubio-Godoy, M. Morphological and molecular description of eight new species of Gyrodactylus von Nordmann, 1832 (Platyhelminthes: Monogenea) from poeciliid fishes, collected in their natural distribution range in the Gulf of Mexico slope, Mexico. Parasitol Res 114, 3337–3355 (2015). https://doi.org/10.1007/s00436-015-4559-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4559-z