Abstract
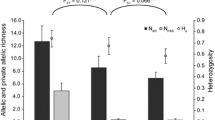
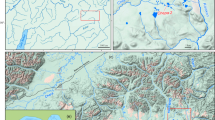
Mechanisms related to ecological or sexual selection have favoured sympatric speciation events in African and Central American lake cichlids. Allopatric divergence is the predominant speciation process observed in Amazonia, although, to the best of our knowledge, no study to date has attempted to determine whether speciation processes could exist under sympatric conditions in Amazonian cichlids. The Apistogramma agassizii species complex is an excellent model for investigating the existence of sympatric divergence events in the Amazon, as it shares many common life history characteristics with African Haplochromine cichlids in which sympatric speciation mechanisms are well documented. The genetic structure of A. agassizii was analysed by genotyping 889 individuals with ten microsatellite loci, collected from 26 sites distributed among small streams in 11 micro-basins in a very small portion of the Peruvian Amazon. It revealed 22 genetic populations identified according to panmictic criteria (FIS estimator) and strongly differentiated: FST estimator (0.034 to 0.356). Such a strong genetic structuring on such small geographical areas has never been demonstrated before in an Amazonian fish. Several of these populations may have diverged sympatrically and repeatedly in small stream networks. The results are discussed with respect to divergence processes, including sympatric speciation, which may be associated with the observed genetic structure.





Similar content being viewed by others
Data Availability
Data are available from the authors upon reasonable request.
References
Albert, J., & Reis, R. (2011). Historical Biogeography of Neotropical Freshwater Fishes. New York: University of California Press. https://doi.org/10.1525/california/9780520268685.001.0001
Amado, M., Farias, I., & Hrbek, T. (2011). A Molecular perspective on systematics, taxonomy and classification amazonian discus fishes of the genus Symphysodon. International Journal of Evolutionary Biology, 2011, 1–16. https://doi.org/10.4061/2011/360654
ArcGIS® software by Esri. ArcGIS® and ArcMap™ are the intellectual property of Esri (1995–2015)
Amado, M., Farias, I., & Hrbek, T. (2011). A molecular perspective on systematics, taxonomy and classification Amazonian discus fishes of the genus Symphysodon. International Journal of Evolutionary Biology, 2011, 360654. https://doi.org/10.4061/2011/360654
Barlow, G. W., & Siri, P. (1997). Does sexual selection account for the conspicuous head dimorphism in the Midas cichlid? Animal Behaviour, 53(3), 573–584. https://doi.org/10.1006/anbe.1996.0332.
Barluenga, M., & Meyer, A. (2004). The Midas cichlid species complex: Incipient sympatric speciation in nicaraguan cichlid fishes? Molecular Ecology, 13(7), 2061–2076. https://doi.org/10.1111/j.1365-294X.2004.02211.x
Barluenga, M., Stolting, K. N., Salzburger, W., Muschick, M., & Meyer, A. (2006). Sympatric speciation in nicaraguan crater lake cichlid fish. Nature, 439, 719–723. https://doi.org/10.1038/nature04325.
Belkhir, K., Borsa, P., Chikhi, L., Raufaste, N., & Bonhomme, F. (2004). Genetix, logiciel sous windows TM pour la génétique des populations. Laboratoire Génome, populations, interactions CNRS UMR 5000. Université de Montpellier II.
Belkhir, K., Castric, V., & Bonhomme, F. (2002). identix, a software to test for relatedness in a population using permutation methods. Molecular Ecology Notes, 2(4), 611–614. https://doi.org/10.1046/j.1471-8286.2002.00273.x.
Bougeard, S., & Dray, S. (2018). Supervised multiblock analysis in R with the ade4 package. Journal of Statistical Software, 86(1), 1–17. https://doi.org/10.18637/jss.v086.i01
Brock, C. D., & Wagner, C. E. (2018). The smelly path to sympatric speciation? Molecular Ecology, 27(21), 4153–4156. https://doi.org/10.1111/mec.14845.
Burress, E. D., Piálek, L., Casciotta, J. R., Almirón, A., Tan, M., Armbruster, J. W., & et, & Říčan, O. (2018). Island- and lake-like parallel adaptive radiations replicated in rivers. Proceedings of the Royal Society B: Biological Sciences, 285(1870), 20171762. https://doi.org/10.1098/rspb.2017.1762
Chanseau, M., Bosc, S., Galiay, E., & Oules, G. (2002). L’utilisation de l’huile de clou de girofle comme anesthésique pour les smolts de saumon atlantique (Salmo salar L) et comparaison de ses effets avec ceux du 2-phenoxyethanol. Bulletin Français de la Pêche et de la Pisciculture. https://doi.org/10.1051/kmae:2002054
Chessel, D., Dufour, A. B., & Thioulouse, J. (2004). The ade4 package-I- one-table methods. R News, 4, 5–10.
Crispo, E., Bentzen, P., Reznick, D. N., Kinnison, M. T., & Hendry, A. P. (2006). The relative influence of natural selection and geography on gene flow in guppies. Molecular Ecology, 15(1), 49–62. https://doi.org/10.1111/j.1365-294X.2005.02764.x.
Dagosta, F., & de Pinna, M. (2019). The fishes of the Amazon: Distribution and biogeographical patterns, with a comprehensive list of species. Bulletin of the American Museum of Natural History, 431(1), 1–163. https://doi.org/10.1206/0003-0090.431.1.1
Danley, P. D., & Kocher, T. D. (2001). Speciation in rapidly diverging systems: Lessons from Lake Malawi. Molecular Ecology, 10(5), 1075–1086. https://doi.org/10.1046/j.1365-294x.2001.01283.x
Deacon, A. E., Jones, F. A. M., & Magurran, A. E. (2018). Gradients in predation risk in a tropical river system. Current Zoology, 64(2), 213–221. https://doi.org/10.1093/cz/zoy004
Dias, M. S., Cornu, J. F., Oberdorff, T., Lasso, C. A., & Tedesco, P. A. (2013). Natural fragmentation in river networks as a driver of speciation for freshwater fishes. Ecography, 36(6), 683–689. https://doi.org/10.1111/j.1600-0587.2012.07724.x.
Dodrill, M. J., Yard, M., & Pine Iii, W. E. (2016). Assessing predation risks for small fish in a large river ecosystem between contrasting habitats and turbidity conditions. American Midland Naturalist, 175(2), 206–221. https://doi.org/10.1674/0003-0031-175.2.206.
Doyle, J. J., & Doyle, J. L. (1987). A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemistry Bulletin, 19, 11–15.
Dray, S., & Dufour, A. B. (2007). The ade4 package: implementing the duality diagram for ecologists. Journal of Statistical Software, 22(4), 1–20. https://doi.org/10.18637/jss.v022.i04.
Dray, S., Dufour, A. B., & Chessel, D. (2007). The ade4 package - II: two-table and K-table methods. R News, 7(2), 47–52.
Elmer, K. R., Lehtonen, T. K., Kautt, A. F., Harrod, C., & Meyer, A. (2010). Rapid sympatric ecological differentiation of crater lake cichlid fishes within historic times. BMC Biology, 8, 60. https://doi.org/10.1186/1741-7007-8-60.
Engelking, B., Römer, U., & Beisenherz, W. (2010). Intraspecific colour preference in mate choice by female Apistogramma cacatuoides HOEDEMAN, 1951 (Teleostei: Perciformes: Cichlidae). Vertebrate Zoology, 60(3), 199–208.
Estivals, G., Duponchelle, F., Römer, U., García-Dávila, C., Airola, E., Deléglise, M., & Renno (2020). The amazonian dwarf cichlid Apistogramma agassizii (Steindachner, 1875) is a geographic mosaic of potentially tens of species: conservation implications. Conservation: Marine and Freshwater Ecosystems, 30(8), 1521–1539. https://doi.org/10.1002/aqc.3373.
Evanno, G., Regnaut, S., & Goudet, J. (2005). Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Molecular Ecology, 14(8), 2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Fitzpatrick, B. M., Fordyce, J. A., & Gavrilets, S. (2008). What, if anything, is sympatric speciation? Journal of Evolutionary Biology, 21(6), 1452–1459. https://doi.org/10.1111/j.1420-9101.2008.01611.x.
Funk, D. J., Nosil, P., & Etges, W. J. (2006). Ecological divergence exhibits consistently positive associations with reproductive isolation across disparate taxa. Proceedings of the National Academy of Sciences of the United States of America, 103(9), 3209–3213. https://doi.org/10.1073/pnas.0508653103.
Gavrilets, S. (2004). Fitness landscapes and the origin of species. Princeton University Press.
Goulding, M., Barthem, R., & Ferreira, E. J. G. (2003). The Smithsonian Atlas of the Amazon. Smithsonian Institution.
Hanfling, B., & Weetman, D. (2006). Concordant genetic estimators of migration reveal anthropogenetically enhanced source-sink population structure in the river sculpin, Cottus gobio. Genetics, 173(3), 1487–1501. https://doi.org/10.1534/genetics.105.054296.
Hubert, N., Duponchelle, F., Nuñez, J., García-Dávila, C., Paugy, D., & Renno, J. F. (2007). Phylogeography of the piranha genera Serrasalmus and Pygocentrus: Implications for the diversification of the neotropical ichthyofauna. Molecular Ecology, 16(10), 2115–2136. https://doi.org/10.1111/j.1365-294X.2007.03267.x
Jombart, T. (2008). Adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics, 24(11), 1403–1405. https://doi.org/10.1093/bioinformatics/btn129.
Junk, W. J., Bayley, P. B., & Sparks, J. S. (1989). In D. P. Dodge (Ed.), The flood pulse concept in river floodplain systems. ( vol. 106). Canadian Special Publication of Fisheries and Aquatic Sciences. pp. 110–127
Junk, W. J., & Wantzen, K. M. (2004). In R. L. Welcomme, & T. Petr (Eds.), The flood pulse concept: new aspects, approaches and applications—An update: vol. Volume I (pp. 117–149). Food and Agriculture Organization and Mekong River Commission, FAO Regional Office for Asia and the Pacific.
Kearse, M., Moir, R., Wilson, A., Stones-Havas, S., Cheung, M., Sturrock, S., Buxton, S., Cooper, A., Markowitz, S., Duran, C., Thierer, T., Ashton, B., Meintjes, P., & Drummond, A. (2012). Geneious basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics, 28(12), 1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kocher, T. D. (2004). Adaptive evolution and explosive speciation: the cichlid fish model. Nature Reviews Genetics, 5(4), 288–298. https://doi.org/10.1038/nrg1316.
Kornfield, I., & Smith, P. F. (2000). African cichlid fishes: model systems for evolutionary biology. Annual Review of Ecology Evolution and Systematics, 31, 163–196. https://doi.org/10.1146/annurev.ecolsys.31.1.163.
Leitão, C. S. S., Santos, C. H. A., Souza, É. M. S., Vilarinho, G. C. C., Paula-Silva, M. N., Val, P., Val, A. L., & de Almeida-Val, V. M. F. (2017). Development and characterization of microsatellite loci in amazonian dwarf cichlids Apistogramma spp. (Perciformes: Cichlidae): Uncovering geological influence on amazonian fish population. Journal of Applied Ichthyology, 33(6), 1196–1199. https://doi.org/10.1111/jai.13490
Maan, M. E., & Seehausen, O. (2010). Mechanisms of species divergence through visual adaptation and sexual selection: perspectives from a cichlid model system. Current Zoology, 56(3), 285–299. https://doi.org/10.1093/czoolo/56.3.285.
Machado-Schiaffino, G., Kautt, A. F., Torres-Dowdall, J., Baumgarten, L., Henning, F., & Meyer, A. (2017). Incipient speciation driven by hypertrophied lips in Midas cichlid fishes? Molecular Ecology, 26(8), 2348–2362. https://doi.org/10.1111/mec.14029.
Malinsky, M., Challis, R. J., Tyers, A. M., Schiffels, S., Terai, Y., Ngatunga, B. P., Miska, E. A., Durbin, R., Genner, M. J., & Turner, G. F. (2015). Genomic islands of speciation separate cichlid ecomorphs in an east african crater lake. Science, 350(6267), 1493–1498. https://doi.org/10.1126/science.aac9927.
Mallet, J. (2008). Hybridization, ecological races, and the nature of species: empirical evidence for the ease of speciation. Philosophical Transactions of the Royal Society of London Series B, 363(1506), 2971–2986. https://doi.org/10.1098/rstb.2008.0081.
Mallet, J., Meyer, A., Nosil, P., & Feder, J. L. (2009). Space, sympatry and speciation. Journal of Evolutionary Biology, 22(11), 2332–2341. https://doi.org/10.1111/j.1420-9101.2009.01816.x.
Mantel, N. (1967). The detection of disease clustering and a generalized regression approach. Cancer Research, 27(2), 209–220.
Marques, D. A., Lucek, K., Haesler, M. P., Feller, A. F., Meier, J. I., Wagner, C. E., Excoffier, L., & Seehausen, O. (2017). Genomic landscape of early ecological speciation initiated by selection on nuptial colour. Molecular Ecology, 26(1), 7–24. https://doi.org/10.1111/mec.13774.
Mathieu, E., Autem, M., Roux, M., & Bonhomme, F. (1990). Épreuves de validation dans l’analyse de structures génétiques multivariées: comment tester l’équilibre panmictique? Revue de Statistique Appliquée, 38, 47–66.
Mayr, E. (1942). Systematics and the origin of species. Harvard University Press.
McGee, M., Borstein, S., Meier, J., Marques, D., Mwaiko, S., Taabu, A., Kishe, M. A., O’Meara, B., Bruggmann, R., Excoffier, L., & Seehausen, O. (2020). The ecological and genomic basis of explosive adaptive radiation. Nature, 586, 75–79. https://doi.org/10.1038/s41586-020-2652-7.
Meier, J. I., Marques, D. A., Mwaiko, S., Wagner, C. E., Excoffier, L., & Seehausen, O. (2017). Ancient hybridization fuels rapid cichlid fish adaptive radiations. Nature Communications, 8, 1–11. https://doi.org/10.1038/ncomms14363.
Nei, M. (1972). Genetic distance between populations. The American Naturalist, 106(949), 283–292. https://doi.org/10.1086/282771.
Oksanen, J., Blanchet, F. G., Kindt, R., Legendre, P., Minchin, P., O’Hara, B., Simpson, G. L., Solymos, P., Stevens, M. H. H., Szoecs, E., & Wagner, H. (2015). Vegan: Community Ecology Package. R Package Version, 2(2-1), 1–2.
Paquette, S. R. (2012). PopGenKit: useful functions for (batch) file conversion and data resampling in microsatellite datasets. R Package Version 1.0. Molecular Ecology Notes, 2, 611–614. https://doi.org/10.1046/j.1471-8278.2002.00273.x.
Piálek, L., Říčan, O., Casciotta, J., Almirón, A., & Zrzavý, J. (2012). Multilocus phylogeny of Crenicichla (Teleostei: Cichlidae), with biogeography of the C. lacustris group: species flocks as a model for sympatric speciation in rivers. Molecular Phylogenetics and Evolution, 62(1), 46–61. https://doi.org/10.1016/j.ympev.2011.09.006.
Poelstra, J. W., Richards, E. J., & Martin, C. H. (2018). Speciation in sympatry with ongoing secondary gene flow and a potential olfactory trigger in a radiation of Cameroon cichlids. Molecular Ecology, 27(21), 4270–4288. https://doi.org/10.1111/mec.14784.
Poulton, E. B. (1904). What is a species? Proceedings of the Entomological Society of London, 1903, lxxvii–cxvi.
Pritchard, J. K., Stephens, M., & Donnelly, P. (2000). Inference of population structure using multilocus genotype data. Genetics, 155(2), 945–959. https://doi.org/10.1093/genetics/155.2.945.
Quérouil, S., Diaz, V., García-Dávila, A., Römer, C., U., & Renno, J. F. (2015). Development and characterization of polymorphic microsatellite markers in neotropical fish of the genus Apistogramma (Perciformes: Labroidei: Cichlidae). Journal of Applied Ichthyology, 31(S4), 52–56. https://doi.org/10.1111/jai.12975.
Rasband, W. S., 1997–2012: ImageJ.US National Institutes of Health, Bethesda.
Ready, J. S., Sampaio, I., Schneider, H., Vinson, C., Santos, D., T., & Turner, G. F. (2006). Colour forms of amazonian cichlid fish represent reproductively isolated species. Journal of Evolutionary Biology, 19(4), 1139–1148. https://doi.org/10.1111/j.1420-9101.2006.01088.x.
Říčan, O., Piálek, L., Dragová, K., & Novák, J. (2016). Diversity and evolution of the middle american cichlid fishes (Teleostei: Cichlidae) with revised classification. Vertebrate Zoology, 66, 1–102.
Rice, W. R., & Hostert, E. E. (1993). Laboratory experiments on speciation: what have we learned in forty years? Evolution, 47(6), 1637–1653. https://doi.org/10.1111/j.1558-5646.1993.tb01257.x.
Richards, E. J., Poelstra, J. W., & Martin, C. H. (2018). Don’t throw out the sympatric speciation with the crater lake water: fine-scale investigation of introgression provides equivocal support for causal role of secondary gene flow in one of the clearest examples of sympatric speciation. Evolution letters, 2(5), 524–540. https://doi.org/10.1002/evl3.78.
Richards, E. J., Servedio, M. R., & Martin, C. H. (2019). Searching for sympatric speciation in the genomic era. Bioessays, 41(7), 1–10. https://doi.org/10.1002/bies.201900047
Rico, C., & Turner, G. F. (2002). Extreme microallopatric divergence in a cichlid species from Lake Malawi. Molecular Ecology, 11(8), 1585–1590. https://doi.org/10.1046/j.1365-294X.2002.01537.x.
Römer, U. (2000). Cichlid atlas. In natural history of south American dwarf cichlids (Vol. 1). Mergus Verlag.
Römer, U. (2006). Cichlid atlas. Natural history of south american dwarf cichlids. (Vol. 2). Mergus Verlag.
Römer, U., & Beisenherz, W. (2005). Intra- and interspecific mate choice of female Apistogramma cacatuoides (Teleostei: Cichlidae). Ichthyological Exploration of Freshwaters, 16(4), 339–345.
Römer, U., Engelking, B., & Beisenherz, W. (2014). Genetically determined mate choice can be influenced by learning in Apistogramma cacatuoides Hoedemann, 1951 (Teleostei, Cichlidae). Vertebrate Zoology, 64(2), 199–206.
Rousset, F. (1997). Genetic differentiation and estimation of gene flow from F-statistics under isolation by distance. Genetics, 145, 1219–1228. https://doi.org/10.1093/genetics/145.4.1219.
Salzburger, W. (2009). The interaction of sexually and naturally selected traits in the adaptive radiations of cichlid fishes. Molecular Ecology, 18(2), 169–185. https://doi.org/10.1111/j.1365-294X.2008.03981.x.
Salzburger, W., & Meyer, A. (2004). The species flocks of east african cichlid fishes: recent advances in molecular phylogenetics and population genetics. Naturwissenschaften, 91, 277–290. https://doi.org/10.1007/s00114-004-0528-6.
Schiesari, L., Zuanon, J., Azevedo-Ramos, C., Garcia, M., Gordo, M., Messias, M., & Vieira, E. (2003). Macrophyte rafts as dispersal vectors for fishes and amphibians in the Lower Solimes River, Central Amazon. Journal of Tropical Ecology, 19(3), 333–336. https://doi.org/10.1017/S0266467403003365.
Schluter, D. (2000). The ecology of adaptive radiation. Oxford University Press.
Seehausen, O., & Magalhaes, I. S. (2010). Geographical Mode and Evolutionary mechanism of ecological speciation in Cichlid Fish. In P. Grant & R. Grant (Eds.), In search of the causes of evolution (pp. 282–308). Princeton University Press.
Seehausen, O., Terai, Y., Magalhaes, I. S., Carleton, K. L., Mrosso, H. D., Miyagi, R., Van der Sluijs, I., Schneider, M. V., Maan, M. E., Tachida, H., Imai, H., -, & Okada, N. (2008). Speciation through sensory drive in cichlid fish. Nature, 455(7213), 620–626. https://doi.org/10.1038/nature07285.
Seehausen, O., Van Alphen, J. J. M., & Witte, F. (1997). Cichlid fish diversity threatened by eutrophication that curbs sexual selection. Science, 277(5333), 1808–1811. https://doi.org/10.1126/science.277.5333.1808.
Seehausen, O., & Wagner, C. E. (2014). Speciation in Freshwater Fishes. Annual review of ecology evolution and systematics, 45(1), 621–651. https://doi.org/10.1146/annurev-ecolsys-120213-091818
Sioli, H. (1984). The Amazon and its main affluents: Hydrography, morphology of the river courses, and river types. In H. Sioli (Ed.), The Amazon: Limnology and landscape ecology of a mighty tropical river and its basin (pp. 127–165). Dr W. Junk Publishers.
Steindachner, F. (1875). Beiträge der Kenntniss der Chromiden des Amazonenstromes. Sitzungsberichte der kaiserlichen Akademie der Wissenschaften. Mathematischnaturwissenschafliche Classe, 71, 61–137.
Van Doorn, G. S., Edelaar, P., & Weissing, F. J. (2009). On the origin of species by natural and sexual selection. Science, 326(5960), 1704–1707. https://doi.org/10.1126/science.1181661.
Van Doorn, G. S., & Weissing, F. J. (2001). Ecological versus sexual selection models of sympatric speciation: A synthesis. Selection, 2(1–2), 17–40. https://doi.org/10.1556/Select.2.2001.1-2
Wagner, C. E., Harmon, L. J., & Seehausen, O. (2012). Ecological opportunity and sexual selection together predict adaptive radiation. Nature, 487(7407), 366–369. https://doi.org/10.1038/nature11144.
Wagner, C. E., McCune, A. R., & Lovette, I. J. (2012). Recent speciation between sympatric Tanganyikan cichlid colour morphs. Molecular Ecology, 21(13), 3283–3292. https://doi.org/10.1111/j.1365-294X.2012.05607.x.
Wahlund, S. (1928). Zusammensetzung von populationen und korrelationserscheinungen vom standpunkt der vererbungslehre aus betrachtet. Hereditas, 11(1), 65–106. https://doi.org/10.1111/j.1601-5223.1928.tb02483.x.
Wallace, A. R. (1876). The geographical distribution of animals. Harper.
Weir, B. S., & Cockerham, C. C. (1984). Estimating F-statistics for the analysis of population structure. Evolution, 38(6), 1358–1370. https://doi.org/10.2307/2408641.
Willis, S. C., Winemiller, K. O., Montaña, C. G., Macrander, J., Reiss, P., Farias, I. P., & Ortí, G. (2015). Population genetics of the speckled peacock bass (Cichla temensis), South America’s most important inland sport fishery. Conservation Genetics, 16, 1345–1357. https://doi.org/10.1007/s10592-015-0744-y.
Wilson, A. B., Noack-Kunnmann, K., & Meyer, A. (2000). Incipient speciation in sympatric Nicaraguan crater lake cichlid fishes: Sexual selection versus ecological diversification. Proceedings of the Royal Society B: Biological Sciences, 267(1458), 2133–2141. https://doi.org/10.1098/rspb.2000.1260
Acknowledgements
Guillain Estivals was supported by a scholarship of the Franco-Peruvian School of Life Sciences. Research was funded by and carried out within the framework of the International Mixt Laboratory - Evolution and Domestication of Amazonian Ichthyofauna (LMI-EDIA), Peruvian Amazon Research Institute - French National Research Institute for Sustainable Development - Gabriel René Moreno Autonomous University (IIAP-IRD-UAGRM). We would like to thank Philippe Cubry from UMR DIADE (Montpellier, France) for his advice on data analysis.
Author information
Authors and Affiliations
Contributions
Conceptualization: Guillain Estivals, Fabrice Duponchelle, Jean-François Renno. Developing methods: GE, FD, J-FR. Conducting the research: GE, FD, J-FR. Data analysis: GE, CM. Data interpretation: GE, FD, CM, J-FR. Preparation figures & tables: GE. Data interpretation, writing: GE, FD, CG-D, UR, CM, J-FR.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Estivals, G., Duponchelle, F., García-Dávila, C. et al. Exceptional Genetic Differentiation at a Micro-geographic Scale in Apistogramma agassizii (Steindachner, 1875) from the Peruvian Amazon: Sympatric Speciation?. Evol Biol 50, 1–17 (2023). https://doi.org/10.1007/s11692-022-09587-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-022-09587-9