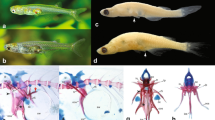
Abstract
The western mosquitofish (Gambusia affinis affinis) is a useful model for the study of sexual dimorphism and the neural circuits associated with sexual differentiation. This is largely because of its anal fin, which undergoes radical postnatal transformation in males. Understanding the neural mechanisms involved in this process may also help elucidate basic principles of the nervous system. The authors describe the mosquitofish as a model for research and present guidelines for the care and use of this species.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Krogh, A. The progress of physiology. Am. J. Physiol. 90, 243–251 (1929).
Krebs, H.A. The August Krogh Principle: “For many problems there is an animal on which it can be most conveniently studied.” J. Exp. Zool. 194, 221–226 (1975).
Krebs, H.A. & Krebs, J.R. The “August Krogh Principle.” Comp. Biochem. Physiol. 67B, 379–380 (1980).
Marder, E. Models identify hidden assumptions. Nat. Neurosci. 3, 1198 (2000).
Marder, E. Non-mammalian models for studying neural development and function. Nature 417, 318–321 (2002).
Baird, S.F. & Girard, G. Descriptions of new species of fishes collected by Mr. J. H. Clark on the U. S. and Mexican boundary survey under Lt. Col. J. D. Graham. Proc. Acad. Nat. Sci. Phila. 6, 390 (1854).
Rosa-Molinar, E., Hendricks, S.E., Rodriguez-Sierra, J.F. & Fritzsch, B. The development of the anal fin appendicular support in the western mosquitofish, Gambusia affinis affinis (Baird and Girard, 1854): a reinvestigation and reinterpretation. Acta Anat. 151, 20–35 (1994).
Rosa-Molinar, E., Fritzsch, B. & Hendricks, S.E. Organizational-activational concept revisited: sexual differentiation in an atherinomorph teleost. Horm. Behav. 30, 563–575 (1996).
Rosa-Molinar, E., Proskocil, B.J., Hendricks, S.E. & Fritzsch, B. A mechanism for anterior transposition of the anal fin and its appendicular support in the western mosquitofish, Gambusia affinis affinis (Baird and Girard, 1854). Acta Anat. 163, 75–91 (1998).
Rosa-Molinar, E. in Viviparous Fishes (eds. Uribe, M.C.A. & Grier, H.J) 66–70 (New Life Publications, Inc., Homestead, FL, 2005).
Holtfreter, J. in The Emergence of Order in Developing Systems: 27th Symposium of the Society for Developmental Biology (ed. Locke, M.) 19–20 (Academic, London, 1968).
Parenti, L.R. A phylogenetic and biogeographic analysis of cyprinodontiform fishes (Teleostei, Atherinomorpha). Bull. Am. Mus. Nat. Hist. 168, 335–557 (1981).
Parenti, L.R. Relationship of atherinomorph fishes (Teleostei). Bull. Mar. Sci. 52, 170–196 (1993).
Rosen, D.E. & Gordon, M. Functional anatomy and evolution of male genitalia in poeciliid fishes. Zoologica 38, 1–52 (1953).
Rosen D.E. & Bailey, R.M. The poeciliid fishes (Cyprinodontiformes) their structure, zoogeography, and systematics. Bull. Am. Mus. Nat. Hist. 126, 1–176 (1963).
Krumholz, L.A. Reproduction of the western mosquitofish, Gambusia affinis (Baird and Girard), and its use in mosquito control. Ecol. Monogr. 18, 1–43 (1948).
Fetcho, J.R., Higashijima S.I. & McLean, D.L. Zebrafish and motor control over the last decade. Brain Res. Rev. 57, 86–93 (2008).
Fetcho, J.R. & Liu, K.S. Zebrafish as a model system for studying neuronal circuits and behavior. Ann. NY Acad. Sci. 860, 333–345 (1998).
Driever, W., Stemple, D., Schier, A. & Solnica-Krezel, L. Zebrafish: genetic tools for studying vertebrate development. Trends Genet. 10, 152–159 (1994).
Rauchenberger, M. Systematics and biogeography of the genus Gambusia (Cyprinodontiformes, Poeciliidae). Amer. Mus. Novit. 2591 (1989).
Parenti, L.R. & Song, J. in Interrelationships of Fishes (eds. Stiassny, M.L.J., Parenti, L.R. & Johnson, G.D.) 427–444 (Academic, San Diego, 1996).
Lauder, G.V. & Liem, H.F. The evolution and interrelationships of the actinopterygian fishes. Bull. Mus. Comp. Zool. 150, 95–197 (1983).
Powers, D.A. Fish as model systems. Science 246, 352–358 (1989).
May, E.B., Bennett, R.O., Lipsky, M.M. & Reimschuessel, R. Using fish as models in biomedical research. Lab Anim. (NY) 16, 23–28 (1987).
May, E.B., Bennett, R.O., Lipsky, M.M. & Reimschuessel, R. Using fish as models in biomedical research. Lab Anim. (NY) 16, 25–31 (1987).
Wolke, R. The use of fish in biomedical research. Comp. Pathol. Bull. 16, 1–6 (1984).
Institute of Laboratory Animal Resources, National Research Council. Guide for the Care and Use of Laboratory Animals 140 (National Academy Press, Washington, DC, 1996).
American Society of Ichthyologists and Herpetologists, American Fisheries Society and American Institute of Research Biologists. Guidelines for use of fishes in field research. Fisheries 13, 16–23 (1988).
DeTolla, L.J. et al. Guidelines for the Care and Use of Fish in Research. ILAR J. 37, 159–173 (1995).
Winterbottom, R. A descriptive synonymy of striated muscles of teleostei. Proc. Acad. Natl. Sci. USA 125, 225–317 (1974).
Rosa-Molinar, E. Proskocil, B.J., Ettel, M. & Fritzsch, B. Whole-mount procedures for simultaneous visualization of nerves, neurons, cartilage, and bone. Brain Res. Prot. 4, 115–123 (1999).
Getting, P.A. Emerging principles governing the operation of neural networks. Annu. Rev. Neurosci. 12, 185–204 (1989).
Wendelaar Bonga, S.E. The stress response in fish. Physiol. Rev. 77, 591–625 (1997).
Wedemeyer, G.A. in Fish Stress and Health in Aquaculture (eds. Iwama, G.K., Pickering, A.D., Sumpter, J.P. & Schreck, C.B.) 35–71 (Cambridge Univ. Press, Cambridge, UK, 1997).
Piper, R.G.G., McCraren, J.P., McElwain, I.B., Orme, L.E. & Fowler, L.G. Fish Hatchery Management 517 (American Fisheries Society, Bethesda, MD 2002).
Tomasso, J.R. in Fish Medicine (ed. Stoskopf, M.K.) 240–246 (WB Saunders, Philadelphia, 1993).
Tucker, C.S. in Fish Medicine (ed. Stoskopf, M.K.) 166–197 (WB Saunders, Philadelphia, 1993).
Winton, J.R. in Fish Hatchery Management 2nd edn. (ed. Wedemeyer, G.A.) 594–611 (American Fisheries Society, Bethesda, MD, 2002).
Stoskopf, M.K. in Fish Medicine (ed. Stoskopf, M.K.) 62–78 (WB Saunders, Philadelphia, 1993).
Stoskopf, M.K. in The Care and Use of Amphibians, Reptiles, and Fish in Research (eds. Schaeffer, D.O., Kleinow, K.M. & Krulisch, L.) 136–141 (Scientists Center for Animal Welfare, Bethesda, MD, 1992).
Thune, R.L., Stanley, L.A. & Cooper, R.K. Pathogenesis of Gram-negative bacterial infections in warmwater fish. Ann. Rev. Fish Dis. 3, 37–68 (1993).
Barrows, F.T. & Hardy, R.W. in Fish Hatchery Management 2nd edn. (ed. Wedemeyer, G.A.) 483–558 (American Fisheries Society, Bethesda, MD, 2002).
Hardy, R.W. in Fish Nutrition (ed. Halver, J.E.) 475–548 (Academic, London, 1989).
Collier, A. The mechanism of internal fertilization in Gambusia. Copeia 1, 45–53 (1936).
Peden, A.E. The function of gonopodial parts and behavioral patterns during copulation by Gambusia. (Poeciliidae). Can. J . Zool. 50, 955–967 (1972).
Peden, A.E. Differences in the external genitalia of female gambusiin fishes. Southwest Nat. 17, 265–272 (1972).
Rose, J.D. The neurobehavioral nature of fishes and the question of awareness and pain. Rev. Fisheries Sci. 10, 1–38 (2002).
Stoskopf, M.K. in Aquaculture for Veterinarians (ed. Brown, L.) 161–167 (Pergamon, New York, 1993).
Summerfelt, R.C. & Smith, L.S. in Methods for Fish Biology (eds. Schreck, C.B. & Moyle, P.B.) 213–272 (American Fisheries Society, Bethesda, MD, 1990).
Matthews, J.L. et al. Pseudoloma neurophilia n. g., n. sp., a new microsporidium from the central nervous system of the zebrafish, Danio rerio. J. Eukaryot. Microbiol. 48, 227–233 (2001).
Kent, M.L. & Bishop-Stewart, J.K. Transmission and tissue distribution of Pseudoloma neurophilia (Microsporidia) of zebrafish, Danio rerio (Hamilton). J. Fish Dis. 26, 423–426 (2003).
Rosen, D. The Care and Breeding of Laboratory Animals 379 (Wiley & Sons, New York, 1950).
American Veterinary Medical Association. AVMA Guidelines on Euthanasia [online] (2007).
Acknowledgements
We thank Jo Taylor for helpful discussions and editorial comments. We thank Aquatic Habitats and Millipore Corporation for past and continued support in the development of the mosquitofish as a laboratory resource. We also thank Nikon Instrument's Nikon Research Alliance Program, specifically Ella de los Santos de Schwartz and Danilo Rosado-Sanchez, for access to cutting-edge imaging technology and technical support. Finally, we acknowledge the contributions of past members of the Biological Imaging Group who are too numerous to mention individually. Portions of this manuscript were presented at the 2006 meeting of the American Association for Laboratory Animal Science. Research in E.R-M.'s laboratory is supported by grants from the National Science Foundation (grant number NSF/IBN-0091120) and the National Institute of Neurological Disorders and Stroke (grant number NIH/5U54 NS30405-07).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Rosario-Ortiz, L., Rivera-Pabon, S., Torres-Vázquez, I. et al. The western mosquitofish (Gambusia affinis affinis): a new laboratory animal resource for the study of sexual dimorphism in neural circuits. Lab Anim 37, 263–269 (2008). https://doi.org/10.1038/laban0608-263
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/laban0608-263